Overview
Design Resources
Design & Integration File
- Host Board
- Sensor Board
- Temperature Flex Board
- Mechanical Files
- Alternative Layout File Formats
Evaluation Hardware
Part Numbers with "Z" indicate RoHS Compliance. Boards checked are needed to evaluate this circuit.
- MAXREFDES104# ($521.73) EV Kit
Description
The MAXREFDES104# is a unique evaluation and development platform in a wearable form factor that demonstrates the functions of a wide range of Maxim’s products for health-sensing applications. This third-generation health sensor platform (an advancement of the MAXREFDES101# Health Sensor Platform 2.0) integrates a two-in-one PPG + ECG analog-front-end (AFE) sensor (MAX86176), a human body temperature sensor (MAX30208), a microcontroller (MAX32666), a power-management IC (MAX20360), and a 3-axis accelerometer. The complete platform includes a 3D-printed enclosure and a biometric algorithm hub with an embedded heart-rate, oxygen saturation and ECG algorithms (MAX32674). Algorithm output and raw data can be streamed through Bluetooth™ to a PC GUI for demonstration, evaluation, and customized development.
Features & Benefits
Features
- Photoplethysmography (PPG)
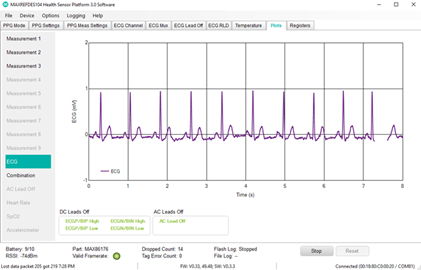
- Biopotential measurement (ECG)
- Skin temperature measurement
- Embedded heart-rate (HRM) and oxygen saturation (SpO2) algorithms
- Motion and acceleration
- Wearable form factor
What's in the Box
- Complete Assembled Wearable
- BLE USB Dongle (CY5677)
- USB-A to USB-C cable
- USB-A to Micro-USB cable
- Programmer Board (MAXDAP-USB-C)
Product Categories
Markets and Technologies
Parts Used
Details Section
The MAXREFDES104# is the third generation in our family of health sensor development platforms. Like the previous generation, MAXREFDES101#, the MAXREFDES104# enables quick evaluation of our latest portfolio of wearable products to significantly shorten the development time for custom fitness, health, and clinical applications.
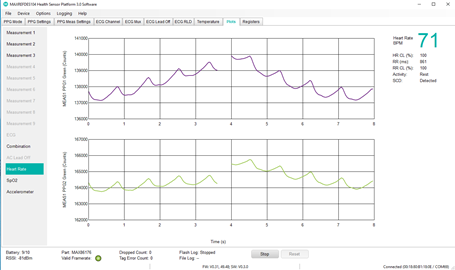
This ready-to-wear wrist form factor reference design monitors blood oxygen saturation (SpO2), electrocardiogram (ECG), pulse rate, heart rate (HR), body temperature and motion. Included algorithms provide HR, heart-rate variability (HRV), respiration rate (RR), SpO2, body temperature, and advanced analytics such as sleep quality and stress level information at clinical-grade levels. This complete reference design can also be used for clinical trials by allowing wearable designers to start collecting data immediately, saving at least six months over building these devices from scratch. Designed for wrist-based form factors, HSP 3.0 can be adapted for other dry electrode form factors such as chest patches and smart rings.
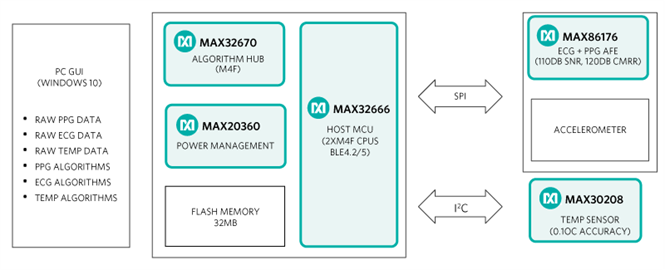
The MAXREFDES104# platform includes the following key components:
- Micro board: Includes the MAX32666 host microcontroller, MAX32674 algorithm and sensor hub, MAX20360 power management IC (PMIC), dual-mode Bluetooth, and 64MB flash storage
- Sensor board: Includes the MAX86176, ECG + PPG AFE, optical heart-rate sensor with three LEDs (Green, Red, and IR), and three photodiodes, stainless steel ECG electrodes, and 3-axis accelerometer
- Temperature flex board: Includes flexible PCB with the MAX30208 body temperature sensor connected to an aluminum disc for optimal skin temperature measurements
- 3D-printed enclosure with adjustable wrist straps
- BLE USB dongle to ensure high data rate
- MAX-DAP-C enables users to reprogram the device with the latest firmware and algorithm libraries
- Design files, hardware, and software
- MAX86176: Lowest-noise optical photoplethysmography (PPG) and electrical ECG analog front end (AFE), which offers 110dB signal-to-noise ratio (SNR) to add SpO2 saturation capability and over 110dB common mode rejection ratio (CMRR) for dry electrode ECG applications. The device enables synchronous acquisition of PPG and ECG measurements, even with independent sample rates, providing pulse transit time for cardiac health use cases.
- MAX20360: Highly integrated power and battery management power management IC (PMIC) optimized for advanced body-worn health sensing devices. It includes Analog Devices Integrated’s high-accuracy ModelGauge™ m5 EZ fuel gauge, a sophisticated haptic driver, and a unique low-noise buck-boost converter that maximizes SNR and minimizes power used for optical bio-sensing.
- MAX32666: Bluetooth (BLE)-enabled, ultra-low power microcontroller with two Arm® Cortex®-M4F cores and an additional SmartDMA which permits running the BLE stack independently, leaving the two main cores available for major tasks. Moreover, the microcontroller integrates an entire security suite and error correcting code (ECC) on the memories to significantly increase the system’s robustness.
- MAX32674: Ultra-low-power microcontroller dedicated to Analog devices Integrated’s world-class PPG algorithms of pulse rate, SpO2, HRV, RR, sleep quality monitoring and stress monitoring. It can be configured either as a sensor hub to support firmware and algorithms or as an algorithm hub to support multiple algorithms. The MAX32674 seamlessly enables customer-desired sensor functionality, including managing the MAX86176 PPG and ECG sensor AFE as well as delivering either raw or calculated data to the outside world.
- MAX30208: The low-power, high-accuracy digital temperature sensor comes in a small package size of 2mm x 2mm. It has 33 percent lower operating current compared to the closest competitive solution. It reads the temperature on the top of the package and can be mounted on a flex cable or PCB, making it easier to design into wearables. With accuracy of 0.1 degrees Celsius, the MAX30208 meets clinical temperature requirements.
You must receive an NDA to order and access the device, software, firmware, and user guide, plus other support collateral such as source code, API documents, example software, and more.
- To apply for the NDA, fill out the request form and agree to the Terms and Conditions.
- Applying for an NDA does not necessarily guarantee approval or shipment of any device or software.
- NDA is subject to approval by the Customer Service Team and will notify applicants accordingly.
- If the NDA is approved, customers will need to register for a myAnalog account (for existing account holders, please log in with your details) in order to access the zip file under the Software Development section (note that this will appear only for approved myAnalog accounts).
- This zip file contains the Windows software GUI, Firmware updates, collateral such as API documents, host source code, and other resources as they become available.
Documentation & Resources
-
MAXREFDES104 Design Files4/20/2022ZIP50 M
-
 MAXREFDES104: Health Sensor Platform 3.012/12/2024
MAXREFDES104: Health Sensor Platform 3.012/12/2024
Support & Training
Search our knowledge base for answers to your technical questions. Our dedicated team of Applications Engineers are also available to answer your technical questions.