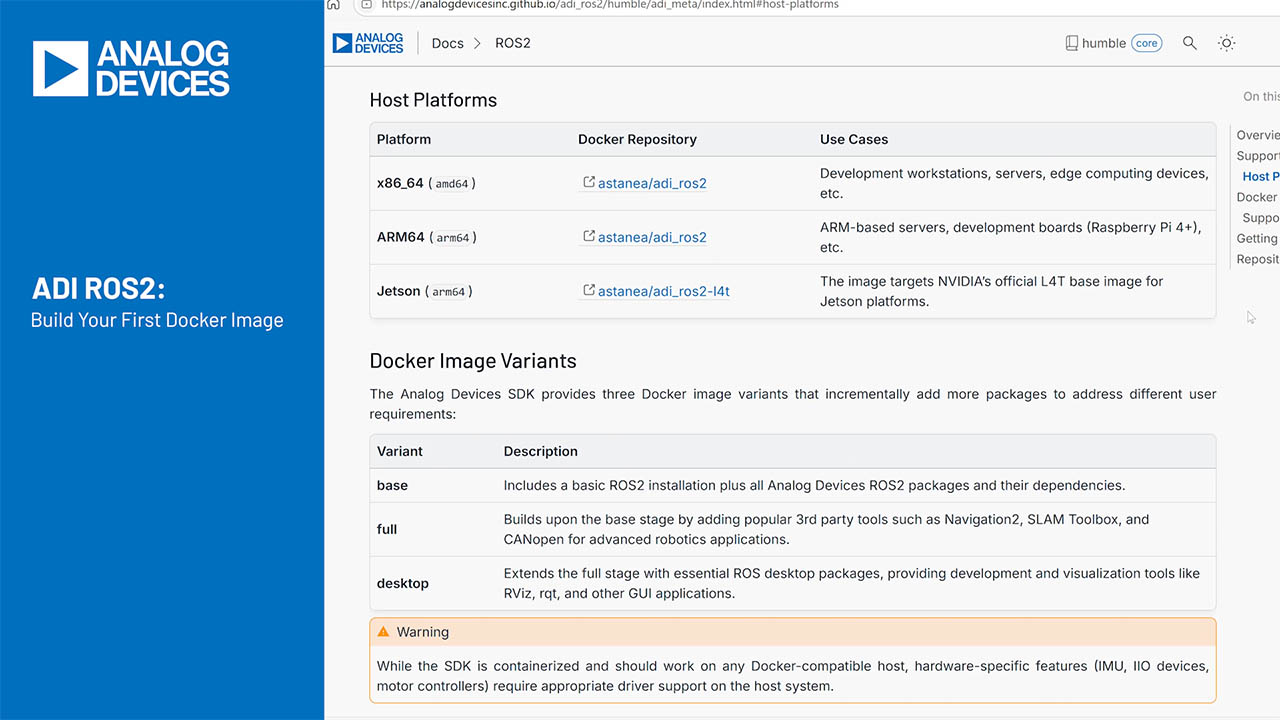
How to Choose the Best Battery for Your Healthcare Medical Device Design
How to Choose the Best Battery for Your Healthcare Medical Device Design
by
Fahad Masood
Apr 17 2024
Abstract
With so many different battery chemistries and form factors, it’s hard to know how to select the appropriate battery for your application. This article will present different criteria to aid engineers in choosing the best battery for their use case. In addition, we’ll discuss five of the most popular primary cell battery chemistries in the market today and applications where they may be a good fit. This article will deal primarily with healthcare applications with guidelines that can be applied to a variety of different products implementing primary cell batteries.
Introduction
Selecting an appropriate primary cell battery can be a balancing act between multiple competing requirements. You want a battery that has enough capacity to power your device for sufficient time and the output voltage range must be adequate for the integrated circuits that you are powering. Typically, you will want as small as possible battery size to minimize overall product dimensions. You’ll want to consider cost, availability, and shelf life. As engineers, it’s also our responsibility to consider the environmental impact of our design decisions. It’s possible that the battery we select for our products could end up in landfills for many years. To help aid the designer’s decision, we’ll focus on the most used alkaline, lithium metal, silver oxide, and zinc air battery chemistries and evaluate them for use in a disposable electrocardiogram (ECG) chest patch design.
Primary vs. Secondary Batteries
The main difference between primary and secondary cell batteries is that primary cell batteries are not rechargeable while secondary batteries are. In primary cell batteries, the electrochemical reaction that occurs is not reversible. Once the anode oxidizes, the battery cannot generate any more electricity. In a rechargeable battery, the anode can be deoxidized. Thus, the battery can be recharged and reused. A secondary battery is typically more expensive than the primary cell battery, which usually precludes it from being used in disposable systems. Primary cell batteries also have a longer shelf life due to their low self-discharge current but rechargeable secondary cell batteries can provide more power, particularly in high current draw applications.
The environmental impact of different types of batteries is a complex issue. On the one hand, secondary batteries are reusable and don’t need to be replaced as often, which means less waste is created. On the other hand, secondary batteries contain hazardous materials that can be harmful to the environment. Primary cell batteries also contain hazardous materials but at a much lower concentration. When comparing the two types of batteries on a per-battery basis, secondary cells discharge more greenhouse gases and create more hazardous waste than primary cell batteries. However, after twenty recharge cycles, secondary batteries produce 90% less waste than disposable primary batteries and are therefore considered to be more environmentally friendly.1
Medical Standards
Batteries for medical applications must meet strict safety and performance standards. The ANSI/AAMI ES 60601-1 standard for medical electrical equipment specifies several regulatory standards that batteries must comply with, including IEC 60086-4 and IEC 60086-5 for primary cell batteries, and UL2054 for household and commercial batteries. In addition, there are specific standards for different applications, such as ISO 20127 for powered toothbrushes.2
The FDA also has specific requirements for lithium batteries, including that they must be produced in a UL-certified factory and that every battery must be traceable for failure analysis. In addition to selecting the correct battery chemistry, it is important to scrutinize the battery manufacturer to ensure that they comply with FDA and IEC regulations for your application.2
Voltage Range
Primary cell batteries are typically available in two voltage ranges, 1.5 V and 3.3 V. The choice of which range to use depends on the application. Buck converters are generally more efficient than boost converters.3 A common strategy with battery regulators is to use a buck-boost converter to maximize the battery voltage range. However, buck-boost converters are usually larger and require more external components than buck converters, since they have four switches instead of two.
| Primary Battery Cell | Min V | Nom V | Max V | Specific Energy |
| Alkaline | 1.1 | 1.5 | 1.65 | 200 Wh/kg |
| Zinc Air | 0.9 | 1.4 | 1.68 | 400 Wh/kg |
| Li Manganese | 2 | 3 | 3.4 | 280 Wh/kg |
| Li Disulfide | 0.9 | 1.5 | 1.8 | 300 Wh/kg |
| Ag Oxide | 1.2 | 1.55 | 1.85 | 130 Wh/kg |

Figure 1. Primary cell battery chemistries.
Alkaline
Alkaline batteries are the most employed primary cell by a considerable margin, partly due to their suitability for powering analog circuits like the ones found in television remote controls or clocks. These batteries have high internal resistance, when compared to other battery chemistries, which increases as the battery discharges. Due to this characteristic, alkaline batteries are not typically suited for digital circuits that require higher loads or have different duty cycles and operating modes. Alkaline cells also exhibit higher internal resistance as the physical size of the cell decreases. Therefore, higher current applications, like a toy with lots of LEDs and speakers, might require a D cell battery, where a clock can run off of a coin cell battery. Alkaline batteries are considered safe to use and store, with minimal concern about explosions or leakage, and are not held to the same regulatory standards as a Li-Ion battery would be.
Alkaline batteries are not typically used for medical devices due to their limited power output and short life span compared to other battery chemistries. In medical applications, they can be found in low cost glucose meters, thermometers, and other devices that are used infrequently and aren’t required for critical functions.

Figure 2. Li-Ion primary cell batteries: lithium manganese dioxide (Li-M or LiMnO2) and also Li-disulfide (Li-FeS2).
There are several lithium-based primary batteries on the market, all of which use lithium as the anode material and a metal as the cathode. These are commonly known as lithium-metal batteries. The two most widely used lithium-metal primary batteries are lithium manganese dioxide (LiMnO2) and lithium disulfide (LiFeS2).
LiMnO2 batteries have a nominal output voltage of 3 V and low internal resistance. This makes them well-suited for digital applications that require different load profiles and duty cycles. LiFeS2 batteries have a nominal output voltage of 1.5 V and similar internal resistance. They are often used as a direct replacement for alkaline batteries in devices that require this voltage.
Lithium-metal batteries are prone to leaking and explosions, so they require special handling and transportation restrictions. However, they offer a number of advantages over alkaline batteries: twice the capacity in similar form factors, a longer lifespan, and lighter weight.
As a result, lithium-metal batteries are replacing alkaline batteries in many applications. Lithium-metal batteries are also used in critical medical devices like continuous glucose meters, infusion pumps, and implantable devices like defibrillators.4
Silver Oxide Batteries
Silver oxide (Ag-O) batteries are another common primary cell battery that uses silver for the cathode and zinc for the anode. They have a similar nominal output voltage with alkaline batteries (that is, 1.55 V) with a higher capacity and flatter discharge curve making them suitable for digital applications. Because of the presence of silver in the cathode, Ag-O batteries can be expensive in large sizes so they’re mostly used in coin cell or button cell form factors.

Figure 3. Silver oxide batteries are commonly used as watch batteries.
Historically, Ag-O batteries have tended to leak, and mercury would be added to the cell to counteract the corrosion. In recent years, battery manufacturers have been able to find other ways to minimize corrosion without the use of mercury, making Ag-O batteries much more sustainable for the environment. Ag-O batteries are generally safer and run longer than lithium batteries while having a similar discharge curve but a higher cost due to the silver cathode limiting their adoption in lower cost applications. Because silver coating can reduce the risk of infections caused by implantable devices, Ag-O battery chemistries are increasingly used in implantable devices.5
Zinc Air
Zinc air batteries have a unique battery chemistry when compared to the previous battery chemistries. Zinc air batteries have a zinc anode and the ambient air is the cathode with an electrolytic paste in between. The cell is constructed in a typical coin cell battery form factor with an opening in the case to allow air in. Before the battery is used, the opening is sealed to prevent air from entering the cell. Once the seal is broken, oxygen is introduced at the cathode and electrons begin their flow from the zinc anode, through the electrolytic paste, and to the cathode. Because the cathode is not a metal, like other battery chemistries, zinc air batteries are lightweight and cost-effective. They also hold their charge and have a relatively flat discharge rate. Zinc air batteries have an output voltage range of 0.9 V to 1.4 V.

Figure 4. Hearing aids are often powered by zinc air batteries.
Because the battery must be exposed to the environment to operate, its usage in medical devices is limited. Many medical devices require a level of protection from the environment, which zinc air batteries do not allow. These chemistries are most used for hearing aid batteries due to their lightweight construction and long life.
Application Example
Now that we have reviewed the common battery chemistries available and what they can offer us, let’s step through an application example. For this example, let’s consider an ECG chest patch with a desired run time of 5 days. This wearable patch will be designed to be disposable, fully sealed (battery cannot be replaced), waterproof, and will have Bluetooth® communication to transmit the ECG data wirelessly. The patch will also include a MAX30208 temperature sensor to record the body temperature of the patient as well as an ADXL367 accelerometer to monitor information about patient activity. It can be used in a hospital setting, an outpatient clinic, as well as at a patient’s home. We know that we want to use the MAX30001as the ECG analog front end (AFE) and the MAX32655 microcontroller unit (MCU) in this application. We will choose the power management solution according to the battery.
Figure 5. An ECG patch example block diagram.
Based on these requirements, we can make an appropriate decision about the battery to use. The wearable design is compact, which means the form factor of the battery should be small and light weight so we should aim to use a coin cell form factor. We can eliminate lithium disulfide batteries as they are not found in coin cell form factors. We know that the patch is disposable so we can’t use a secondary or rechargeable battery. We also know that the battery is fully sealed, which precludes us from using a zinc air cell. Bluetooth communication and the different operating modes of the MAX32655 also lead us to reasonably assume that an alkaline battery could not support this application due to its high internal resistance. That leaves us with lithium manganese and silver oxide primary cell battery chemistries as our options.
The lithium manganese battery has a nominal output voltage of 3.0 V and a higher specific energy than silver oxide batteries. We can easily source a (silver oxide) CR2032 battery that has a capacity of 235 mAh. The silver oxide battery has a nominal output voltage of 1.55 V and the biggest coin cell form factor we can find off the shelf is an SR44W battery with a capacity of 200 mAh. If we go back to our design and look at the requirements, we see that we need a 5 day run time for our patch. By constructing a load profile (discussed in more depth in a previous article6), we find that the patch will consume an estimated 45 mA per day or 225 mA over 5 days. The requirement of a higher capacity battery eliminates the silver oxide coin cell from contention and leaves us with a lithium manganese battery as our choice for this application.
Conclusion
Choosing a battery for your application requires careful consideration of form, fit, and function. By understanding the strengths and weaknesses of each battery chemistry, you can select the best battery for your system design requirements.
References
1 Giovanni Dolci, Camilla Tua, Mario Grosso, and Lucia Rigamonti. “Life CycleAssessment of Consumption Choices: A Comparison Between Disposable andRechargeable Household Batteries.” The International Journal of Life Cycle Assessment, Vol. 21, 2016.
2 Jeff Shepard. “What’s Different About Industrial and Medical Li Batteries?” Battery Power Tips, 2023.
3 “Prolong Your Smart Watch Lifetime with SIMO.” Analog Devices, Inc., 2019.
4 David C Bock, Amy Marschilok, Kenneth J. Takeuchi, Esther S. Takeuchi. “Batteries Used to Power Implantable Biomedical Devices.” Electrochim Acta, Vol. 84, 2012.
5 J. M. Schierholz, L. J. Lucas, A. Rump, and G. Pulverer. “Efficacy of Silver-Coated Medical Devices.” The Journal of Hospital Infection, Vol. 40, 1998.
6 Fahad Masood. “Power Supply Design Considerations for Primary Cell Powered Remote Patient Monitors.” Analog Devices, Inc., April 2022.
About the Authors
Fahad Masood is a senior applications engineer for health and medical biosensing applications at Analog Devices. He has nearly a decade of experience in electronics development for healthcare, computing, and industrial app...
Related to this Article
Products
PRODUCTION
±0.1°C Accurate, I2C Digital Temperature Sensor
PRODUCTION
Ultra-Low-Power, Single-Channel Integrated
Biopotential (ECG, R-to-R, and Pace Detection)
and Bioimpedance (...
RECOMMENDED FOR NEW DESIGNS
Low-Power, Arm Cortex-M4 Processor with FPU-Based Microcontroller and Bluetooth 5.2