概览
设计资源
评估硬件
产品型号带"Z"表示符合RoHS标准。评估此电路需要下列选中的电路板
- EVAL-ADICUP3029 ($60.92) Ultra Low Power Arduino form factor compatible Development Platform
- EVAL-CN0428-EBZ ($67.68) Water Quality Sensor pH or Conductivity
- EVAL-M355-ARDZ-INT ($67.68) ADuCM355 based Sensor Interposer Board
优势和特点
- 测量1个(最多4个)传感器通道
- 可选SPI、I2C或UART通信
- 10引脚JTAG/SWD连接器,便于编程
- 尺寸兼容Arduino
参考资料
-
CN0428 User Guide2018/11/9WIKI
-
Solutions For Rapid Prototyping: Answering the Needs of Practicing Engineers2023/8/23PDF784 K
-
CN0428: 水质测量系统2018/11/13PDF434 K
-
液体测量—从水到血液2019/10/1
-
YOU DRINK WHAT?!? Water Quality Analysis for Environmental Measurement2020/9/25
-
面向环境和过程测量的智能检测2019/11/14
电路描述
pH测量的基本原理
pH值是衡量水溶液中氢离子和氢氧化物离子相对量的一项指标。就摩尔浓度来说,25°C的水含有 1 × 10−7 摩尔/升氢离子,氢氧化物离子浓度与此相同。中性溶液指氢离子浓度正好等于氢氧化物离子浓度的溶液。pH值是表示氢离子浓度的另一种方式,定义如下:

因此,如果氢离子浓度为 1.0 × 10−2 摩尔/升,则pH值为2.00。
pH电极是许多工业所使用的电化学传感器,但对水处理和污水工业具有特别重要的意义。pH探针由一个玻璃测量电极和一个参比电极构成,类似于一块电池。当把探针置于溶液中时,测量电极产生一个电压,具体取决于溶液中氢的活性,然后将该电压与参比电极的电位进行比较。随着溶液酸性的增强(pH值变低),玻璃电极电位相对于参比电极阳性增强(+mV)。随着溶液碱性的增强(pH值变高),玻璃电极电位相对于参比电极阴性增强(-mV)。这两个电极之差即为测得电位。在理想情况下,典型的pH探针在25°C下会产生±59.154 mV/pH电压,用能斯特方程表示为:

其中:
E为氢电极电压,活性未知。
a = ±30 mV,零点容差。
T 为环境温度 (°C)。
n = 1 (25°C时) ,化合价(离子上的电荷数)。
F = 96485 库仑/摩尔,法拉第常数。
R = 8.314 伏特-库仑/°K摩尔,理想气体常数。
pH 为未知溶液的氢离子浓度。
pHISO 为参比氢离子浓度。请参阅探针文档。典型 pHISO = 7。
方程表明,产生的电压取决于溶液的酸碱度,并以已知方式随氢离子活性而变化。溶液温度的变化会改变其氢离子的活性。当溶液被加热时,氢离子运动速度加快,结果导致两个电极间电位差的增加。相反,当溶液冷却时,氢活性降低。理想情况下,根据设计,当置于pH值为7的缓冲溶液中且 pHISO 为7时,电极产生零伏电位。
pH值测量中的温度补偿
当测量溶液的pH值时,须考虑的重要参数之一是温度变化。当溶液的温度改变时,溶液的pH值也会发生可观的变化。这个改变的值不是pH读数的误差,而是新温度下溶液的真实pH值。
温度变化可能导致测量电极的灵敏度发生变化,进而引起测量误差。该误差是可预测的,并且可通过全温度范围内的探针校准和后续测量期间的温度校正来解决。
理想电极是在pH 7下精确归零的电极。在pH 7时,温度对电极灵敏度的影响可以忽略不计。然而,大多数pH电极不是理想电极,存在由于温度变化而导致的电极灵敏度问题。一般温度误差非常接近0.003 pH/℃/偏离pH 7的pH单位数。在这种情况下,必须校准pH计以应用此0.003的校正因子。
用校准好的温度传感器,即可实现这种补偿。然后,该温度传感器便能告知温度的变化(如有)。如果存在变化,则将“0.003 pH/°C/偏离pH 7的pH单位数”的校正因子读数输入最终pH读数,pH计将能显示经过校正且更准确的读数。该机制能够很好地补偿由于温度变化引起的pH值误差。
pH校准
由于电极涂层和老化原因,pH电极的特性会随时间而变化。因此,需要使用校准程序来获得更高精度。
校准通过测量两种缓冲溶液的pH值来完成,各缓冲溶液的pH值已知。软件包括不同pH值缓冲溶液的NIST查找表,以及0°C至95°C温度校正的pH值。溶液温度利用电阻温度检测器(RTD)测得。使用以下线性等式:

确定pH传感器转换函数的实际斜率,测量实际失调电压。为了计算斜率,需求解下式:

其中:
y1 为第一点的测量电压。
y2 为第二点的测量电压。
x1 为第一点的已知pH值。
x2 为第二点的已知pH值。
进行上述测量并将一个校准点代入等式2,便可根据以下最终等式确定未知pH值:

其中:
x 为未知pH值。
y 为测量电压。
b 为测量失调电压。
m 为斜率。
然后可以使用等式3来调整先前所述能斯特方程给出的值。
更多信息请查阅CN-0428用户指南。
电导率测量基础知识
电导率衡量物质传导电流的能力高低。物质允许电流流动的程度是其电导率的量度。金属和半导体的导电性随着带电粒子的移动而发生。在有外加电位差的情况下,原子外壳中的价电子移动,从而引起电子的流动,产生电流。
纯水只有很少量的H2O分子离解为H+ 离子和OH− 离子,因此电导率非常低,但在大多数日常情况下遇到的水中则含有相当数量的离子物质,导致电导率提高。自来水、蒸馏水、地表水、工业设备用水等等,具有不同的离子组分。因此,测量溶液的电导率需要测量溶液中存在的这些离子体的行为。
溶液中存在的离子组分具有正电荷或负电荷。当通过外部电极(阴极和阳极)在这种溶液中施加电位差时,这些电极之间的阳离子和阴离子的移动会引起电流。对产生电流的离子流的测量,就是对该溶液的电导率的测量。
电导率在很大程度上取决于溶液的多种因素,包括迁移率、化合价、离子浓度以及溶液温度。而且,电导率与溶液的纯度高度相关。例如,超纯水样不含许多离子成分,因此电导率低,约为5 μS/cm。而含有金属盐和离子化合物的水样则包含许多可导电的阳离子和阴离子,因此电导率值较大。盐水大致为5 S/cm。这种差异使得电导率测量的动态范围很宽。
与金属和半导体的情况一样,溶液的电导率遵守欧姆定律。当在两个电极之间施加一个电位差(V)时,随着阳离子和阴离子的移动,电极之间发生电流(I)的流动。
该电压V与电流I成正比:V α I。

比例常数就是溶液的电阻(R),可以使用欧姆定律计算,如下式所示。电阻单位为欧姆(Ω)。

其中:
V 为电压,单位为伏特(V)。
I 为电流,单位为安培(A)。
R 为溶液的电阻,单位为欧姆(Ω)。
电阻的倒数称为电导(G),用西门子(S)来衡量,等于 Ω−1。

电化学中的另一个重要参数是电导池常数(K),它是电极之间的距离(d)与电极的面积(a)之比。

其中:
K 为电导池常数 (cm−1)。
a 为电极的有效面积 (cm2)。
d 为电极之间的距离 (cm)。
总结先前的计算,电导率是溶液传导电流的能力,等于电导与电导池常数的乘积。电导率(κ)定义如下:

其中:
G = 电导 (S)
K = 电导池常数 (cm−1)
因为电导池常数K用 (cm−1)表示,所以电导率(ρ)通常用S/cm表示,大多数环境水的电导率在mS/cm或μS/cm范围内。

ORP测量基础知识
氧化还原电位(ORP)是对物质获得电子(氧化)或失去电子(还原)的倾向的电化学测量。它是一种溶液氧化或还原另一种化学物质的特性,单位为毫伏。在同时含有氧化剂和还原剂的溶液中,ORP取决于氧化剂与还原剂的相对数量和强度。ORP和氧化还原传感器均很常用,可以互换。实际测量与pH值测量类似。pH值是通过测量氢离子活性来表示酸/碱水平,氧化还原测量则是通过测量电子活性来表示溶液的氧化/还原能力。结果为正值的ORP读数总是表示氧化环境,而负ORP值表示还原环境。ORP表明特定反应预期是否可能发生。通过了解ORP以及溶液的各种其他参数,例如溶液的pH值、温度和杂质,便可对预期发生的反应进行详细分析。
虽然ORP的测量与pH值的测量相似,但正确选择电极是二者的区别所在。必须使用低电阻的电极。优选电极通常是铂或金等惰性金属电极。
在电解过程中,这些低电阻电极要么向氧化剂释放电子,要么不断从还原剂接受电子,直至达到平衡状态。由于电极上电荷的累积,最终会实现稳定状态,其产生的电压就是溶液的ORP。
溶液的氧化还原电位部分地是温度的函数,这反映在能斯特方程中。
ORP电极根据能斯特半电池电位方程测量氧化还原电位,如下所示:

其中:
E 为测得的电极电位。
E0 为所分析系统的特定电压。 对于特定反应, E0 的值可在化学手册中查到。
T 为绝对温度 (K)。
n 为氧化物质和还原物质达成平衡所涉及的电子数。
F = 96,485 库仑/摩尔,法拉第常数。
R = 8.314 伏特-库仑/°K摩尔,理想气体常数。
Aox 为氧化剂的活性。
ARed 为还原剂的活性。
电化学模拟前端
ADuCM355为电化学测量提供一种平台解决方案,其中集成了所有必要的测量功能以及一个低功耗微处理器(MCU)。除了主要测量和电化学功能外,还可以利用任意测量功能进行定制和灵活的测量。因此,它是一款完整的测量平台,尺寸很小,功耗极低,溶液可重复使用以检测多个参数,并且可在传感器外壳内部实现,而功能和性能堪比台式仪器。ADuCM355的高集成度可减少器件数量,提高可靠性,并提供比分立实施解决方案更一致的性能。
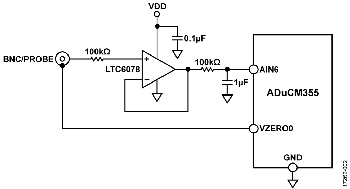
微功耗精密放大器
LTC6078能够为pH探针等高阻抗传感器提供精密缓冲。其精密性能和低偏置电流(输入偏置电流最大值为1 pA)是此设计的关键因素,其中pH探针的阻抗可能有数千兆欧(GΩ)。
LTC6078的低1/f噪声系数(1μVp-p)是低速测量系统实现高测量精度的另一个重要特性。LTC6078在低电源电压下保持精密性能,具有高输入阻抗和宽增益带宽,以超低功耗(54μA)工作,并具有关断功能。
应用模式
EVAL-CN0428-EBZ板支持pH值和阻抗两种测量模式及温度补偿,具体取决于EVAL-CN0428-EBZ板开关和跳线的配置。表1显示了每种测量的各种开关配置。
开关S1有两个设置,允许用户选择pH或Z设置,印刷电路板(PCB)上有相应的标记。用户可以使用高阻抗LTC6078缓冲器来测量电压(使用pH设置),或使用ADuCM355恒电位仪来测量阻抗或电导率(使用Z设置)。
开关S2用于选择阻抗测量范围:ADuCM355内部电阻(INT)或LTC6078提供的10 MΩ跨阻放大器(TIA)范围。对于高速TIA (HSTIA),ADuCM355内部电阻最高可达128 kΩ;对于低功耗TIA (LPTIA),内部电阻最高可达512 kΩ。 EVAL-CN0428-EBZ还有一个位于ADuCM355外部的1 MΩ电阻,用于阻抗测量期间电流范围较低的情况。
| 期望测量 | S1设置 | S2设置 |
| pH | pH | 不适用 |
| 电导率或阻抗,在100 Ω至10 MΩ范围内自动调整量程 | Z | INT选择ADuCM355内部电阻,自动调整量程 |
| 大于200 kΩ的低电流电导率或阻抗 | Z | HI-Z选择10 MΩ LTC6078低电流TIA范围 |
pH模式注意事项
为使用户能够利用高阻抗LTC6078缓冲器测量电压,必须将开关S1设置为pH。pH模式适用于任何电压输出传感器,包括pH、ORP或其他电位传感器,如离子选择性电极。
阻抗模式注意事项
要将EVAL-CN0428-EBZ设置为阻抗模式,必须将开关S1设置为Z。阻抗模式适用于电导率传感器、电化学阻抗谱扫描或任何其他基于电阻或电流的测量。
当S2处于INT模式时(采用内部ADuCM355 TIA范围和自动调整量程),用户可以测量从<100 Ω到大约10 MΩ的阻抗。
当测量阻抗或电导率时,ADuCM355上的测量固件会自动调整TIA及可编程增益放大器(PGA)的增益,以使输入信号最大化,同时不让模数转换器(ADC)饱和。这会大大增加测量的动态范围,从而可以测量宽范围的电导率,以及ORP等低电阻传感器和pH探针等高电阻传感器的阻抗。为了改善如此宽范围内的精度,确实需要多个外部校准电阻。这些额外的校准电阻包含在设计中。如果电路必须改变范围,测量时间会增加。这种增加在低频测量时是很明显的。
当S2处于高阻模式时(采用外部10 MΩ范围和LTC6078),用户可以测量200 kΩ至>100 MΩ的阻抗。S2是一种机械开关,漏电流更低,这也意味着在10 MΩ高阻抗范围内无法自动调整量程。可检测的最大电流约为100 nA。因此,这种模式下无法检测200 kΩ以下的阻抗。
传感器连接
BNC连接器用于主传感器测量,可以是pH值、ORP或任何2线电导率探针。并非所有水质探针都是基于BNC的,但它是模拟输出探针最常用的连接器。
RCA连接器用于温度测量。温度测量常常通过负温度系数(NTC)热敏电阻集成到探针本身之中。PT100和PT1000等RTD温度传感器也很常见。RCA是一种常用接口,但还有其他接口,例如香蕉插头。
当没有RCA插头或RCA适配器时,可以使用接头P3来测量温度输入。
常见变化
建议不要将四电极电导率探针直接与EVAL-CN0428-EBZ一起使用。但是,ADuCM355一般可以支持这些探针。如果终端系统中需要4电极测量,请参阅M355_4WireImpedance示例项目,其位于ADuCM355软件库中。
电路评估与测试
该电路使用EVAL-CN0428-EBZ评估板(见图5)、EVAL-ADICUP3029基于Arduino的无线开发平台(见图6)以及EVAL-M355-ARDZ-INT Arduino扩展接口板(见图7)。有关平台板的用户指南,请访问www.analog.com/EVAL-ADICUP3029。
该系统由EVAL-ADICUP3029板、EVAL-M355-ARDZ-INT Arduino扩展板(插入EVAL-ADICUP3029板)和最多四个插入Arduino扩展板的EVAL-CN0428-EBZ传感器子板组成。EVAL-ADICUP3029Cortex M3与Arduino兼容,充当主机MCU。其功能是与EVAL-CN0428-EBZ子板(其与传感器接口)上的测量引擎进行通信。
Arduino扩展板是EVAL-ADICUP3029和传感器板之间的接口。一次最多可以将四个传感器板安装到扩展板上。水质传感器子板展示了ADuCM355与BNC和RCA连接器接口以支持现成的pH值、电导率、ORP和温度探针。
设备要求
需要以下设备:
- EVAL-ADICUP3029 电路板。
- EVAL-M355-ARDZ-INT Arduino兼容平台。
- 加载有固件的EVAL-CN0428-EBZ 水质传感器板(最多四个)。
- 具有USB端口和 Windows® 7 (32位)或更高版本的PC。
- 串行终端软件(PuTTY、Tera Term或类似软件)。
- USB A型转micro-USB电缆。
- 测试溶液。
- BNC 连接器探针(最多四个)
- RCA 连接器温度探针(可选,最多四个)
- 烧杯(可选)
- 探针支架(可选)
- 用于探针校准的校准缓冲液(可选)
- 外部调试器或额外的EVAL-ADICUP3029板,以便对子板进行重新编程而不切割EVAL-ADICUP3029(可选)上的走线。有关对子板上的ADuCM355重新编程的更多信息,请参阅用户指南。
推荐设备
建议使用以下探针:
- Cole-Parmer 100系列替换pH值/温度电极。
- Cole-Parmer 100系列替换电导率/温度探针,K = 1。
- Sensorex S550C-ORP重型ORP传感器。




设置
设置电路进行评估的步骤如下:
- 将最多四个EVAL-CN0428-EBZ板插入EVAL-M355-ARDZ-INT扩展板,然后插入EVAL-ADICUP3029电路板。
- 确保EVAL-M355-ARDZ-INT和EVAL-CN0428-EBZ评估板上的开关设置正确。
- 将EVAL-ADICUP3029虚拟COM USB端口连接到PC。
- 设置串行终端软件以匹配CN-0428固件的设置,并选择正确的虚拟COM端口
- 按下EVAL-ADICUP3029板上的复位按钮,软件显示水质测量步骤。
子板附带专用固件,但也提供了条件来供用户编写新固件。子板可由EVAL-ADICUP3029的调试器部分通过USB进行编程。但是,这需要切割三条走线并使用附带的电缆连接到EVAL-M355-ARDZ-INT。切断这些走线后,需要使用电缆来通过USB进行通信。其他方案是使用额外的EVAL-ADICUP3029,或使用外部调试器。详细说明请参阅CN-0428用户指南。
有关硬件和软件操作的详细信息,请参阅CN-0428用户指南;该文档可在ADI公司网站上找到,请访问www.analog.com/CN0428-UserGuide。

性能结果
该板有四个校准范围,专门用于TIA提供的不同增益。这些是具有可编程增益的内部TIA。图10显示了在校准后不同增益范围内测量已知电阻值时的百分比误差。该图展现了系统测量已知阻抗的精度。通过校准电阻来补偿开关电阻和ADuCM355开关矩阵的漏电,可以有助于提高阻抗的测量精度。各个板可以进一步校准以获得更好的精度。如需更多信息,请参阅CN-0428用户指南。

然后使用传感器板,在室温下测试瓶装水的pH值和电导率,从而评估其性能。图11、图12和图13分别是针对pH值、电导率和ORP获得的示例图。





