Linear Charger for Nickel Cadmium or Nickel Metal Hydride Batteries Minimizes Parts Count
Linear Charger for Nickel Cadmium or Nickel Metal Hydride Batteries Minimizes Parts Count
著者
Fran Hoffart
2005年09月01日
Introduction
Although rechargeable Lithium Ion and Lithium Polymer batteries have lately been the battery of choice in high performance portable products, the old workhorse nickel cadmium (NiCd) and the newer nickel metal hydride (NiMH) batteries are still important sources of portable power. Nickel based batteries are robust, capable of high discharge rates, good cycle life and are relatively inexpensive. NiMH batteries are replacing NiCd in many applications because of the higher capacity ratings (40 to 50% higher) and because of environmental concerns of the cadmium contained in NiCd cells. This article covers NiCd/NiMH battery charging basics, and introduces the LTC4060 linear battery charger.
A Primer on Charging NiCd/NiMH Batteries
The various methods for charging Nickel based batteries are categorized by speed: slow, quick and fast. The simplest type of charger is a slow charger, which applies a timer-controlled, relatively low charge current for about 14 hours. This may be too long for many portable applications. For shorter charge times, quick and fast chargers apply a constant current while monitoring the battery voltage and/or temperature to determine when to terminate or stop the charge cycle. Charge times typically range anywhere from 3 to 4 hours (quick charge) to around 0.75–1.5 hours (fast charge).
Fast and quick chargers force a constant charge current and allow the battery voltage to rise to the level it requires (within limits) to force this current. During the charge cycle, the charger measures the battery voltage at regular intervals to determine when to terminate the charge cycle. During the charge cycle, the battery voltage rises as it accepts charge (see Figure 1). Near the end of the charge cycle, the battery voltage begins to rise much faster, reach a peak, then begins to fall. When the battery voltage has dropped a fixed number of mV from the peak (–ΔV), the battery is fully charged and the charge cycle ends.
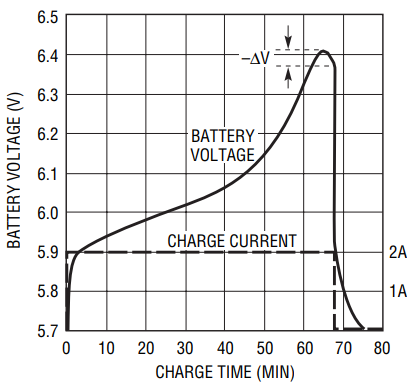
Figure 1. Typical charge profile for a 2000mAHr NiMH 4-cell battery pack charged at a 1C rate.
The battery has an internal safeguard against overcharge. While the cell voltage is dropping from its peak, the battery temperature and internal pressure quickly rise. If fast charging continues for a significant amount of time after full charge is reached, the battery pressure seal may momentarily open causing gas to vent. This is not necessarily catastrophic for the battery, but when a cell vents, some electrolyte is also released. If venting occurs often, the cell will eventually fail. In addition, after venting, the seal may not close correctly and the electrolyte can dry out.
Differences Between NiCd and NiMH Batteries
The open circuit voltage (nominal 1.2V) and the end-of-life voltage (0.9V to 1V) are almost identical between the two battery types, but the charging characteristics differ somewhat. All NiCd cells can be trickle charged continuously, but some NiMH cells cannot, and may be damaged if the trickle charge is continued after reaching full charge. Also, the battery voltage profile during a fast charge cycle differs between the two battery types.
For NiMH cells, the decrease in battery voltage (–ΔV) after reaching a peak is approximately one half that of NiCd cells, thus making charge termination based on –ΔV slightly more difficult. In addition, the NiMH battery temperature rise during the charge cycle is higher than NiCd, and the higher temperature further reduces the amount of –ΔV that occurs when full charge is reached. For NiMH cells, –ΔV is almost non-existent at high temperatures for charge rates less than C/2. (See sidebar for the definition of “C”). Older batteries and cell mismatching further reduce the already minute drops in battery voltage.
Other differences between the two chemistries include higher energy density and greatly reduced voltage depression or “memory effect” for NiMH cells, although NiCd is still preferred for high current drain applications. NiCd cells also enjoy lower self-discharge characteristics, but NiMH technology has room to improve in this regard, while NiCd technology is fairly mature.
The LTC4060 NiCd/NiMH Battery Charger Controller
The LTC4060 is a complete NiCd or NiMH linear battery charger controller that provides a constant charge current and charge termination for fast charging up to four series-connected cells. Simple to use and requiring a minimum of external components, the IC drives an inexpensive external PNP transistor to provide charge current. The basic configuration requires only five external components, although additional functions are included such as, NTC input for battery temperature qualification, adjustable recharge voltage, status outputs capable of driving an LED and shutdown and pause inputs. Selecting the battery chemistry and the number of cells to charge is accomplished by strapping pins, and the charge current is programmed using a standard value resistor. With adequate thermal management, charge current up to 2A is possible, and even higher current when using an external current sense resistor in parallel with the internal sense resistor.
What’s Important When Designing a Charger Using the LTC4060?
Once the battery chemistry and number of cells are set, one must determine the correct charge current. The LTC4060 is designed for fast charging nickel-based batteries and uses –ΔV as the charge termination method. Battery temperature can also be monitored to avoid excessive battery temperature during charging, and a safety timer shuts down the charger if charge termination does not occur. The typical fast charge voltage profile (the rapid rise, then drop in battery voltage (–ΔV) near the end of the charge cycle) only occurs at a relatively high charge current. If the charge current is too low, the battery voltage does not produce the required drop in battery voltage after reaching a peak, which is necessary for the LTC4060 to terminate the charge cycle. At very low charge current, –ΔV does not occur at all. On the other hand, if the charge current is too high, the battery may become excessively hot requiring an NTC thermistor located near the battery to pause the charge cycle allowing the battery to cool down before resuming the charge cycle.
A Typical LTC4060 Charge Cycle
With sufficient input voltage applied, no battery connected and the correct charge current, charge time and thermistor connections in place, the charger’s output voltage is very close to the input voltage. Connecting a discharged battery to the charger pulls down the charger’s output voltage below 1.9 • VCELL (VCELL is the total battery voltage divided by the number of cells being charged) thus starting a charge cycle.
If the battery temperature, as measured by the NTC thermistor, is outside a 5°C to 45°C window, the charge cycle pauses and no charge current flows until an acceptable temperature is reached. When the battery temperature is within limits, the battery voltage is measured and must be below the max limit.
If VCELL is below 900mV, the charger begins a trickle charge of 20% of the programmed charge current until the voltage exceeds 900mV, at which point the full programmed charge current begins. Several hundred milliseconds after the charge cycle begins, if the battery voltage exceeds 1.95V, the charge cycle stops. This overvoltage condition usually means the battery is defective requiring that the charger be manually reset by replacing the battery, toggling the shutdown pin, or removing and reapplying power.
Once the programmed constant charge current starts flowing, a period of time known as “hold-off-time” begins. This hold-off-time ranges from 4 minutes to 15 minutes depending on the charge current and charge time settings. During the hold off time, the –ΔV termination is disabled to prevent false charge termination. A battery that is deeply discharged or has not been charged recently may exhibit a drop in battery voltage during the early portion of the charge cycle, which could be mistaken for a valid –ΔV termination.
During the charge cycle, the battery voltage slowly rises. When the battery approaches full charge, the battery voltage begins to rise faster, reaches a peak, then begins to drop. The charger continuously samples the battery voltage every 15 to 40 seconds, depending on charge current and timer settings. If each sampled voltage reading is less than the previous reading, for four consecutive readings, and the total drop in battery voltage exceeds 8mV/cell for NiMH or 16mV/cell for NiCd, the charge current stops, ending the charge cycle. The open drain output pin “CHRG”, which was pulled low during the charge cycle, now becomes high impedance.
A user programmable recharge feature starts a new charge cycle if the battery voltage drops below a set voltage level because of self-discharge or a load on the battery. Also, if a fully charged battery greater than 1.3V is connected to the charger, the –ΔV termination detection circuit is enabled immediately with no hold-off-time, thus shortening the charge cycle for a battery that is already close to full charge.
If the battery reaches approximately 55°C during the charge cycle, the charger pauses until the temperature drops to 45°C, then resumes charging until the –ΔV termination ends the charge cycle. If no –ΔV termination takes place, the safety timer stops the charge cycle. If the timer stops the charge cycle, it is considered a fault condition and the charger must be reset by removing and replacing the battery, toggling the SHDN pin or toggling the input power to the charger.
About Battery Capacity and Charge Current
The correct charge current is always related to a battery’s capacity, or simply “C”. The letter “C” is a term used to indicate the manufacturers stated battery discharge capacity, which is measured in mA • Hr. For example, a 2000mAHr rated battery can supply a 2000mA load for one hour before the cell voltage drops to 0.9V or zero capacity. In the same example, charging the same battery at a C/2 rate would mean charging at 1000mA (1A).
The correct charge current for fast charging NiCd or NiMH batteries is between approximately C/2 and 2C. This current level is needed for the cell to exhibit the required –ΔV inflection that occurs when the cell reaches full charge, although charging at 2C may cause excessive battery temperature rise, especially with small, high capacity NiMH cells. Because of chemical differences between the two battery chemistries, NiMH cells generate more heat when fast charging.
Watch Out for These Pitfalls
Don’t connect a load directly to the battery when charging. The charge current must remain relatively constant for the –ΔV charge termination to be effective. Loads with changing current levels result in small changes in battery voltage which can trigger a false –ΔV charge termination. For applications that require a load, refer to the power path components shown in Figure 2. When the input voltage is present, the load is powered from the input supply through Schottky diode D1 and the battery is isolated from the load. Removing the input voltage pulls the gate of Q2 low, turning it on providing a low resistance current path between the battery and the load.
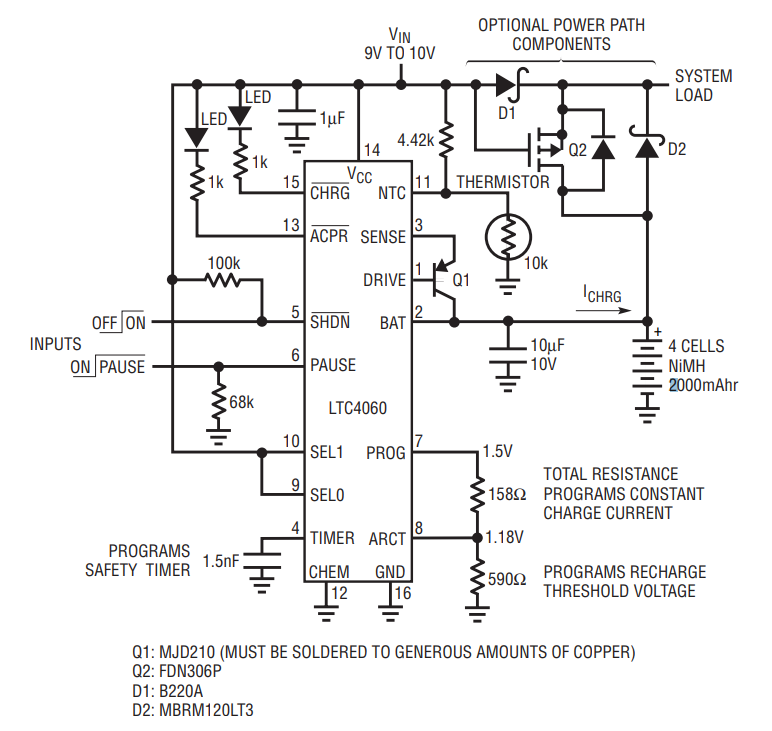
Figure 2. 4-cell 2A NiMH battery charger with NTC thermistor and power path control
Minimize the DC resistance between the charger and the battery. Some battery holders have springs and contacts that have excessive resistance. The increased resistance in series with the battery can prevent a charge cycle from starting because of a battery overvoltage condition once the full charge current begins. Poorly constructed battery holders can also produce false charge termination if battery movement generates a premature –ΔV reading.
Unlike Lithium Ion cells that can be paralleled for increased capacity, NiCd or NiMH cells should not be paralleled, especially when fast charging. Interaction between the cells prevents proper charge termination. If more capacity is required, select larger cells.
Not all NiCd or NiMH batteries behave the same when charging. Manufacturers differ in materials and construction resulting in somewhat different charge voltage profiles or amount of heat generated. A battery can be designed for general purpose use, or optimized for high capacity, fast charge rate, or high temperature operation. Some batteries may not be designed for high current (2C) charge rates resulting in high cell temperature when charging. Also, most new cells are not completely formed and require some conditioning before they reach their rated capacity. Conditioning consists of multiple charge and discharge cycles.
A thermistor mounted near the battery pack, preferably making contact with one or more of the cells, is highly recommended, both as a safety feature and to increase battery lifetime. Unlike Lithium Ion batteries that exhibit very little temperature rise when charging, Nickel based batteries will heat up during the charge cycle, especially NiMH batteries. Minimizing the length of time the battery is exposed to elevated temperature extends battery lifetime.
Conclusion
NiCd and NiMH batteries are ideal sources of rechargeable power for many portable products and backup applications. This article helps to familiarize the user with some of the charging characteristics of nickel based batteries and how they apply to the LTC4060 charger. Charging NiCd and NiMH batteries correctly and safely is simplified using the LTC4060 linear battery charger controller.